Professional Electric Foot Buffer Manufacturer | Bulk OEM Supplies_9
Professional Electric Foot Buffer Manufacturer | Bulk OEM Supplies
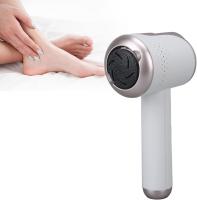
Electric foot buffers revolutionize foot care routines across European spas and Middle Eastern salons. These professional devices eliminate manual filing fatigue while ensuring consistent results. Our solutions address cracked heels and rough skin with clinical precision.
How to Choose the Right Electric Foot Buffer?
Consider motor power, attachment variety, and noise levels. Professional models feature variable speed controls and ceramic rollers.
Technical Comparison: Traditional vs Advanced Models
| Feature | Traditional Buffer | Our Premium Model |
|---|---|---|
| Motor Power | 15W | 35W |
| Noise Level | 65dB | 45dB |
| Attachments | 2 standard | 6 specialized |
| Certification | Basic CE | CE+ISO+RoHS |
Manufacturing Process Excellence
 Our production follows strict SOPs with automated testing at each stage. Component assembly undergoes torque verification and electrical safety checks.
Our production follows strict SOPs with automated testing at each stage. Component assembly undergoes torque verification and electrical safety checks.
Quality Control System
All buffers pass 72-hour endurance testing and insulation resistance verification. We maintain DNV-certified quality management with batch traceability.
Global Logistics and Customization
MOQ: 100 units for standard models. Delivery: 15 days for stock items, 30 days for customized orders. We handle CITES documentation and provide HS code assistance.
Client Testimonials
"The buffer's durability impressed our chain of German spas" - RelaxZone GmbH, Germany. "Monthly order volume increased by 200 units after customer feedback" - BeautyHouse, UAE.
Risk Management
We offer TT/L/C payments with quality guarantee clauses. All shipments include spare parts and operation manuals.
Cross-Border Procurement Tips
Schedule meetings during 9:00-17:00 CET. Prepare import licenses and cosmetic device registration documents. Request sample evaluations before bulk orders.
Call to Action
Download product catalog → Request free sample → Schedule video consultation
Serving 35 countries with annual exports exceeding 500,000 units. All products comply with EU Medical Device Regulation 2017/745.